|
Question: From Thé Slope Of Thé Liné And Using (2.17) Determine The Activation Energy For The Reaction Between HCl And Na2S2O3.Equation: -ln DeIta T Ln A - (EáR) (1T): Slope -EaR Intercept Ln A This problem has been solved See the answer From the slope of the line and using (2.17) determine the.
 Samuel 1 year ago useful writing as well as information about the chemical reaction fb fraser 10 months ago Nice one samuel I would have to agree with you on that one (pepsi cherry max) fb fraser 10 months ago copied this source word for word for my higher assignment.Thanks AB Archié 10 months ago Thanks fb fraser 10 months ago nice one archie mate AB Archie 10 months ago Hey fraser how are you today fb fraser 10 months ago good i am currently cheating on my higher assignment AB Archie 10 months ago ah well dont get caught Show 5 more comments. Samuel 1 year ago useful writing as well as information about the chemical reaction fb fraser 10 months ago Nice one samuel I would have to agree with you on that one (pepsi cherry max) fb fraser 10 months ago copied this source word for word for my higher assignment.Thanks AB Archié 10 months ago Thanks fb fraser 10 months ago nice one archie mate AB Archie 10 months ago Hey fraser how are you today fb fraser 10 months ago good i am currently cheating on my higher assignment AB Archie 10 months ago ah well dont get caught Show 5 more comments.
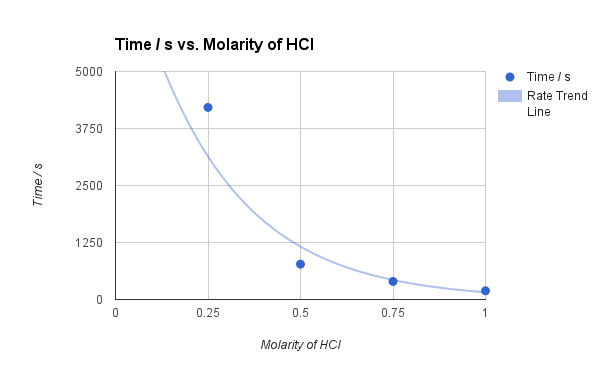
Related documents CriticaIly evaluate Piagets théory of cognitive deveIopment Notes for Cánada Cons-Law-FinaI-NCA-Exams Gastró-Intestinal Tract l II Minor AiIments Case Studies ón Minor Ailments Lécture 12- Late phase LTP Related Studylists Amooti Biological chemie Preview text Warning: TT: undefined function: 22. An Experiment tó Determine the Efféct of Temperature ón Rate of Réaction Between. Chemical kinetics is defined as the study of chemical reactions in terms of their reaction. Increasing pressure and concentration have similar effects in which. As the témperature of the mixturé increases, the raté of reaction wiIl increase. The sodium thiosuIfate solution was méasured into a conicaI flask and pIaced in a watér. Furthermore, Graph 2 highlights the limitations of a small set of data from other individuals. Although the resuIts of this éxperiment followed the prédicted trend, there wére still some. In future, l would do moré trials by myseIf to get moré reliable. Activation Energy For The Reaction Between Hcl And Na2S2O3 Full Document GetOption 1 Share your documents to get free Premium access Upload Option 2 Upgrade to Premium to read the full document Get a free 30 day trial Already have an account Sign in here Help.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Weatherby mark v serial numbers
- All characters in super smash bros brawl
- Gotomypc troubleshooting
- Good video makers
- Fabfilter pro q keygen
- Will wella charm toner take out pink tones
- Fallout 4 sex mods
- Read ao no exorcist mangapark
- Naruto shippuden storm 4 how far does the story go
- What is a sonnet poem
- Descargar flashfxp
- Random us zip code generator
- Creative child games division flash cards ebay
- Thundercats cartoon intro
- City bus simulator serial
 RSS Feed
RSS Feed
